Advertisements
Advertisements
Which nitrogen containing compound amongst the following would undergo Mendius reduction to furnish primary amine \[\ce{(R - NH2)}\]?
Concept: undefined >> undefined
In Dow process, chlorobenzene is converted into phenol by ____________.
Concept: undefined >> undefined
Advertisements
Stock notations are used to specify the oxidation numbers of ____________.
Concept: undefined >> undefined
+I effect of alkyl groups in alcohols increases the stability of ____________.
Concept: undefined >> undefined
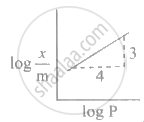
Adsorption of a gas follows Freundlich adsorption isotherm. x is the mass of the gas adsorbed on mass m of the adsorbent. The plot of log `"x"/"m"`· versus log P is shown in the given m graph. `"x"/"m"` is proportional to
Concept: undefined >> undefined
The CORRECT relationship between heat of fusion (∆fusH), heat of vapourization (∆vapH) and heat of sublimation (∆subH) is ____________.
Concept: undefined >> undefined
____________ will NOT undergo acetylation.
Concept: undefined >> undefined
The sum of oxidation states of all atoms in \[\ce{SnO^{2-}_2}\] and CO2 are ____________ respectively.
Concept: undefined >> undefined
Which of the following reactions is appropriate for converting benzamide to aniline?
Concept: undefined >> undefined
For a given mass of an ideal gas, which of the following statements is CORRECT?
Concept: undefined >> undefined
Phenoxide ion is more stable than phenol due to the ____________.
Concept: undefined >> undefined
When methane undergoes combustion in air, the oxidation number of C ____________.
Concept: undefined >> undefined
Which of the following is INCORRECT regarding phenol?
Concept: undefined >> undefined
A gas absorbs 250 J of heat and expands from 1 dm3 to 10 dm3 at constant temperature against external pressure of 0.5 bar. The values of Q, W and ∆U will be respectively ____________.
Concept: undefined >> undefined
Sodium benzene sulphonate reacts with NaOH and then on acidic hydrolysis, it gives __________.
Concept: undefined >> undefined
The most resistant alcohol towards oxidation reaction is:
Concept: undefined >> undefined
Which of the following influence(s) the solubility of a solid in a liquid solvent?
Concept: undefined >> undefined
Henry's law constant for a gas CH3Br is 0.159 mol dm−3 atm−1 at 25°C the solubility of CH3Br in water at 25°C and a partial pressure of 0.164 atm ____________.
Concept: undefined >> undefined

Identify reagents X, Y and Z.
Concept: undefined >> undefined
Which of the following is INCORRECT?
Concept: undefined >> undefined
