Advertisements
Advertisements
What type of hybridization is observed in interhalogen compounds of the type \[\ce{XX^'_3}\]?
Concept: undefined >> undefined
Resonance is NOT exhibited by ____________.
Concept: undefined >> undefined
Advertisements
How many pi bonds and sigma bonds are present in following molecule?
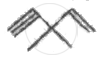
Concept: undefined >> undefined
Which among the following has the lowest surface tension?
Concept: undefined >> undefined
Which of the following statements is not correct?
Concept: undefined >> undefined
Brown ring test is used for detection of which radical?
Concept: undefined >> undefined
Which of the following is not the permissible arrangement of electrons in an atom?
Concept: undefined >> undefined
The most probable product in the reaction given below is:

Concept: undefined >> undefined
Which element among the following does form pπ - pπ multiple bonds?
Concept: undefined >> undefined
In assigning R - S configuration, which among the following groups has highest priority?
Concept: undefined >> undefined
The correct statement with regard to \[\ce{H^+_2}\] and \[\ce{H^-_2}\] is:
Concept: undefined >> undefined
Which element among the following does not form diatomic molecules?
Concept: undefined >> undefined
Identify the group that exerts electron withdrawing resonance effect.
Concept: undefined >> undefined
Identify the functional group that has an electron-donating inductive effect.
Concept: undefined >> undefined
In the case of R, S configuration the group having the highest priority is ______.
Concept: undefined >> undefined
Arrange the following free radicals in order of decreasing stability.
- Methyl
- Vinyl
- Allyl
- Benzyl
Concept: undefined >> undefined
Which of the following element do not follow Aufbau principle?
Concept: undefined >> undefined
Which one of the following laws will represent the pairing of electrons in a subshell after each orbital is filled with one electron?
Concept: undefined >> undefined
Energy required to dissociate 6 g of gaseous hydrogen into free gaseous atoms is 208 kcal at 25°C. Bond energy of H-H bond will be ____________.
Concept: undefined >> undefined
The CORRECT order of C-X bond polarity is ____________.
Concept: undefined >> undefined
