Advertisements
Advertisements
Boric acid (H3BO3) is ______.
Concept: undefined >> undefined
Hydrogen has three isotopes: protium (1H), deuterium (2H or D) and tritium (3H or T). They have nearly the same chemical properties but different physical properties. They differ in ______.
Concept: undefined >> undefined
Advertisements
\[\begin{array}{cc}\phantom{......}\ce{OH}\\
\phantom{....}|\\\ce{CH3CH2 - C - CH3}\\
\phantom{....}|\\\phantom{.....}\ce{Ph}\end{array}\] cannot be prepared by ______.
Concept: undefined >> undefined
Borazine, also known as inorganic benzene, can be prepared by the reaction of 3-equivalents of “X” with 6-equivalents of “Y”. “X” and “Y”, respectively are ______.
Concept: undefined >> undefined
The reaction of H3N3B3Cl3(A) with LiBH4 in tetrahydrofuran gives inorganic benzene (B). Further, the reaction of (A) with (C) leads to H3N3B3(Me)3. Compounds (B) and (C) respectively, are ______.
Concept: undefined >> undefined
The correct decreasing order of priority for the functional groups of organic compounds in the IUPAC system of nomenclature is ______.
Concept: undefined >> undefined
Polar stratospheric clouds facilitate the formation of ______.
Concept: undefined >> undefined
The structures of beryllium chloride in the solid state and vapour phase, are ______.
Concept: undefined >> undefined
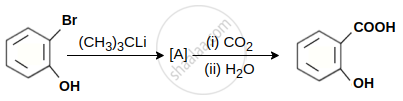
In the given conversion the compound A is:
Concept: undefined >> undefined
Which of the following compound does not contain sulphur atom?
Concept: undefined >> undefined
Which of the following are isostructural pairs?
(A) \[\ce{SO^{2-}4}\] and \[\ce{CrO^{2-}4}\]
(B) SiCl4 and TiCl4
(C) NH3 and \[\ce{NO^-3}\]
(D) BCl3 and BrCl3
Concept: undefined >> undefined
Which of the following compound gives pink colour on reaction with phthalic anhydride in cone. H2SO4 followed by treatment with NaOH?
Concept: undefined >> undefined
The metal that can be purified economically by fractional distillation method is ______.
Concept: undefined >> undefined
Which of the following reaction DOES NOT involve Hoffmann bromamide degradation?
Concept: undefined >> undefined
Given below are two statements:
Statement I: Both CaCl2.6H2O and MgCl2.8H2O undergo dehydration on heating.
Statement II: BeO is amphoteric whereas the oxides of other elements in the same group are acidic.
In the light of the above statements, choose the correct answer from the options given below:
Concept: undefined >> undefined
The functions of antihistamines are ______.
Concept: undefined >> undefined
The internal energy (in J) when 90 g of water undergoes complete evaporation at 100°C is ______.
(Given ΔHvap for water at 373 K = 41 kJ/mol, R = 8.314 JK−1 mol−1)
Concept: undefined >> undefined
Methyl amine on reaction with chloroform in the presence of NaOH gives ______.
Concept: undefined >> undefined
The specific rate constant of the decomposition of N2O5 is 0.008 min−1. The volume of O2 collected after 20 minutes is 16 ml. The volume that would be collected at the end of the reaction is ______ mL. NO2 formed is dissolved in CCl4.
Concept: undefined >> undefined
How many conformations does ethane have?
Concept: undefined >> undefined
