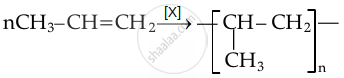Advertisements
Advertisements
Which one of the following is the correct statement?
Concept: undefined >> undefined
Which one of the following cannot function as an oxidizing agent?
Concept: undefined >> undefined
Advertisements
Some drugs bind to a site other than the active site of an enzyme. This site is known as ______.
Concept: undefined >> undefined
If you spill a chemical toilet cleaning liquid on your hand, your first aid would be ______.
Concept: undefined >> undefined
Match List I with List II.
| List - I | List - II | ||
| a. | Siderite | I. | FeCO3 |
| b. | Malachite | II. | CuCO3 · Cu(OH)2 |
| c. | Sphalerite | III. | ZnS |
| d. | Calamine | IV. | ZnCO3 |
Choose the correct answer from the option given below.
Concept: undefined >> undefined
Given below are two statements.
Statement I: In CuSO4·5H2O, Cu-O bonds are present.
Statement II: In CuSO4·5H2O, ligands coordinating with Cu(II) ion are O- and S-based ligands.
In light of the above statement, choose the correct answer from the options given below.
Concept: undefined >> undefined
The gas released during anaerobic degradation of vegetation may lead to ______.
Concept: undefined >> undefined
4.5 g of compound A (MW = 90) was used to make 250 mL of its aqueous solution. The molarity of the solution in M is x × 10−1. The value of x is ______. (Rounded-off to the nearest integer)
Concept: undefined >> undefined
250 mL of 0.5 M NaOH was added to 500 mL of 1 M HCl. The number of unreacted HCl molecules in the solution after complete reaction is ______ × 1021. (Nearest integer) (NA = 6.022 × 1023).
Concept: undefined >> undefined
The equivalents of ethylene diamine required to replace the neutral ligands from the coordination sphere of the trans-complex of CoCl3.4NH3 is ______. (Round off to the Nearest Integer).
Concept: undefined >> undefined
A 6.50 molal solution of KOH (aq.) has a density of 1.89 g cm−3. The molarity of the solution is ______ mol dm−3. (Round off to the Nearest Integer)
[Atomic masses: K: 39.0 u; O: 16.0 u; H: 1.0 u]
Concept: undefined >> undefined
On heating compound (A) gives a gas (B) which is constituent of air. The gas when treated with H2 in the presence of catalyst gives another gas (C) which is basic in nature, (A) should not be ______.
Concept: undefined >> undefined
The following data was obtained for chemical reaction given below at 975 K.
\[\ce{2NO(g) + 2H2(g) -> N2(g) + 2H2O(g)}\]
| [NO] | [H2] | Rate | |
| Mol L-1 | Mol L-1 | Mol L-1 s-1 | |
| (1) | 8 × 10-5 | 8 × 10-5 | 7 × 10-9 |
| (2) | 24 × 10-5 | 8 × 10-5 | 2.1 × 10-8 |
| (3) | 24 × 10-5 | 32 × 10-5 | 8.4 × 10-8 |
The order of the reaction with respect to NO is ______. (Integer answer)
Concept: undefined >> undefined
Assertion (A): Ozone is destroyed by CFCs in the upper stratosphere.
Reason (R): Ozone holes increase the amount of UV radiation reaching the earth.
Concept: undefined >> undefined
The following ligand is:

Concept: undefined >> undefined
Total sodium ions which are present in one formula unit of sodium ethane-1, 2-diaminetetraacetatochromate (II) and sodium hexanitrito cobaltate (III) are ______.
Concept: undefined >> undefined
The correct order of the spin-only magnetic moment of metal ions in the following low spin complexes,
\[\ce{[V(CN)6]^{4-}, [Fe(CN)6]^{4-}, [Ru(NH3)6]^{3+} and [Cr(NH3)6]^2}\] is:
Concept: undefined >> undefined
The major Product of the following reaction is:
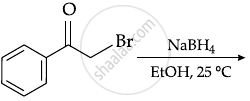
Concept: undefined >> undefined
A drop of solution (volume 0.05 ml) contains 3.0 × 10-6 mole of H+. If the rate constant of disappearance of H+ is 1.0 × 107 mole l-1s-1. It would take for H+ in drop to disappear in ______ × 10-9s.
Concept: undefined >> undefined