Advertisements
Advertisements
Given, that `"E"_("O"_2//"H"_2"O")^circ` = + 1.23 V;
`"E"_("S"_2"O"_8^{2-}//"SO"_4^(2-))` = 2.05 V;
`"E"_("Br"_2//"Br")^circ` = 1.09 V;
`"E"_("Au"^{2+}//"Au")^circ` = + 1.4 V
The strongest oxidizing agent is:
Concept: undefined >> undefined
In which of the following reaction, H2O2 is behaving as a reducing agent:
Concept: undefined >> undefined
Advertisements
The missing structures A and B in the reaction sequence:
\[\ce{R-CH2-CH2OH ->[Al2O3][350°C] R-CH=CH2 ->[(i) O3][(ii) ZnH3O^⊕] RCHO + A;}\]
\[\ce{RCHO ->[Reduce] B;}\]
are:
Concept: undefined >> undefined
Boron compounds behave as Lewis acids because of their ______.
Concept: undefined >> undefined
In which delocalization of positive charge is possible:
Concept: undefined >> undefined
Ge (II) compounds are powerful reducing agents, whereas Pb (IV) compounds are strong oxidants. It can be due to ______.
Concept: undefined >> undefined
If there were 10 periods in a periodic table then maximum number of elements it can have is ______.
Concept: undefined >> undefined
Which of the following reactions of alkanols does not involve \[\ce{C - O}\] bond breaking?
Concept: undefined >> undefined
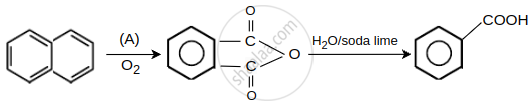
Oxidizing agent (A) used is ______.
Concept: undefined >> undefined
\[\ce{Phenol ->[(i) NaOH][(ii) CO2/140°C] A ->[H+/H2O] B ->[Al2O3][CH3COOH] C}\]
In this reaction, the end product C is ______.
Concept: undefined >> undefined
Sea water is found to contain 5.85% NaO and 9.50% MgCl2 be weight of solution. The normal boiling point of sea water is ______°C assuming 80% ionisation for NaO and 50% ionisation of \[\ce{MgCl2 [Kb(H2O)}\] = 0.51 kg mole-1 K].
Concept: undefined >> undefined
In the following pairs, the one in which both transition metal ions are colourless is ______.
Concept: undefined >> undefined
The transition metal ions responsible for colour in ruby and emerald are, respectively:
Concept: undefined >> undefined
To prepare 3-ethylpentane-3-ol, the reactants needed are ______.
Concept: undefined >> undefined
Hydrogen has three isotopes: protium (1H), deuterium (2H or D) and tritium (3H or T). They have nearly the same chemical properties but different physical properties. They differ in ______.
Concept: undefined >> undefined
Similarity in chemical properties of the atoms of elements in a group of the periodic table is most closely related to ______.
Concept: undefined >> undefined
The correct decreasing order for the metallic character is ______.
Concept: undefined >> undefined
The compound that cannot act both as oxidizing and reducing agent is ______.
Concept: undefined >> undefined
Copper becomes green when exposed to moist air for a long period. This is due to ______
Concept: undefined >> undefined
A group 13 element ‘X’ reacts with chlorine gas to produce a compound XCl3. XCl3 is electron deficient and easily reacts with NH3 to form \[\ce{Cl3X –> NH3}\] adduct; however, XCl3 does not dimerize X is ______.
Concept: undefined >> undefined
