Advertisements
Advertisements
The effective atomic number (EAN) of a metal carbonyl, m(CO)x is 36. The atomic number of the metal is 26. The value of 'x' is ______.
Concept: undefined >> undefined
The de Broglie wavelength of an electron in the 4th Bohr orbit is ______.
Concept: undefined >> undefined
Advertisements
Among the following substituted silanes, which one will give rise to cross linked silicone polymer on hydrolysis?
Concept: undefined >> undefined
The metal salts formed during softening of hard water using Clark's method are ______.
Concept: undefined >> undefined
Permanent hardness in water cannot be cured by ______.
Concept: undefined >> undefined
Match List - I with List - II.
| List I (Polymer) |
List II (Used in) |
||
| (A) | Bakelite | (I) | Radio and television Cabinets |
| (B) | Glyptal | (II) | Electrical switches |
| (C) | PVC | (III) | Paints and Lacquers |
| (D) | Polystyrene | (IV) | Water Pipes |
Choose the correct answer from the options given below:
Concept: undefined >> undefined
Kjeldahl's method was used for the estimation of nitrogen in an organic compound. The ammonia evolved from 0.55 g of the compound neutralised 12.5 mL of 1 MH2SO4 solution. The percentage of nitrogen in the compound is ______. (Nearest integer)
Concept: undefined >> undefined
A sample of 0.125 g of an organic compound when analyzed by Duma's method yields 22. 78 mL of nitrogen gas collected over KOH solution at 280 K and 759 mm Hg. The perentage of nitrogen in the given organic compound is ______. (Nearest integer)
Given:
- The vapour pressure of water of 280 K is 14.2 mm Hg.
- R = 0.082 L atm K-1 mol-1
Concept: undefined >> undefined
The transformation occurring in Duma's method is given below:
\[\ce{C2H7N + (2x + \frac{y}{2}) CuO -> xCO2 + \frac{y}{2} H2O + \frac{z}{2} N2 + (2x + \frac{y}{2}) Cu}\]
The value of y is ______. (Integer answer)
Concept: undefined >> undefined
17.0 g of NH3 completely vapourises at –33.42°C and 1 bar pressure and the enthalpy change in the process is 23.4 kJ mol–1. The enthalpy change for the vapourisation of 85 g of NH3 under the same conditions is ______ kJ.
Concept: undefined >> undefined
According to the valence bond theory, the hybridization of central metal atom is dsp2 for which one of the following compounds?
Concept: undefined >> undefined
Which of the following reagent is used for the following reaction?
\[\ce{CH3CH2CH3 ->[?] CH3CH2CHO}\]
Concept: undefined >> undefined
Number of amphoteric compounds among the following is:
BeO, BaO, Be(OH)2, Sr(OH)2
Concept: undefined >> undefined
At 20°C, the vapour pressure of benzene is 70 torr and that of methyl benzene is 20 torr. The mole fraction of benzene in the vapour phase at 20°C above an equimolar mixture of benzene and methylbenzene is ______ × 10-2. (Nearest integer)
Concept: undefined >> undefined
The major product formed in the following reactions is:
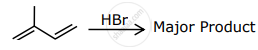
Concept: undefined >> undefined
Which one of the following methods is most suitable for preparing deionized water?
Concept: undefined >> undefined
In carius method of estimation of halogen 0.172 g of an organic compound showed presence of 0.08 g of bromine. Which of these is the correct structure of the compound?
Concept: undefined >> undefined

Select schemes A, B, C out of
(I) acid catalysed hydration
(II) HBO
(III) oxymercuration-demercuration
Concept: undefined >> undefined
What would be the main product when propene reacts with HBr?
Concept: undefined >> undefined
The most stable free radical among the following is:
Concept: undefined >> undefined
