Advertisements
Advertisements
A solution is prepared by mixing 250 ml toluene (C7H8) and 8.4 g thiophene (C4H4S). Then molality of thiophene in the solution is ______. [Given : Density of toluene = 0.8 g/ml, Density of thiophene = 1.2 g/ml]
[Multiply your answer by 10]
Concept: undefined >> undefined
A gem dichloride is formed in the reaction except ______.
Concept: undefined >> undefined
Advertisements
Lauryl alcohol is obtained from coconut oil and is used to make detergent. A solution of 5 g of Lauryl alcohol in 200 g of benzene freezes at 4°C. The approximate molar mass of Lauryl alcohol will be ______.
Given, Kf of benzene = 5.1°C/molal, Freezing point of benzene = 5.5°C.
[Give your answer after division by 17]
Concept: undefined >> undefined
A gaseous mixture was passed at the rate of 2.5 L/min through a solution of NaOH for a total of 1 hour. The SO2 in the mixture was retained as sulphite ion:
\[\ce{SO2(g) + 2OH- -> SO3^2 - (g) + H2O(l)}\]
After acidification with HCl the sulphite was titrated with 5 ml of 0.003 M KIO3
\[\ce{IO_3^- + 2H2SO3 + 2Cl- -> 2SO^{2-}_4 + ICl^-_2 + 2H+ + H2O}\]
The concentration of SO2 will be ______ ppm if density of gaseous mixture is 1.6 gm/L.
Concept: undefined >> undefined
\[\ce{N2 + 3H2 <=> 2NH3 }\]
1 mole N2 and 3 mole H2 are present at start in 1L flask. At equilibrium NH3 formed required 100 mL of 5M HCl for neutralisation hence KC is ______.
Concept: undefined >> undefined
When SO2 is passed through acidified K2Cr2O7 solution ______.
Concept: undefined >> undefined
When 2-alkyne is treated with sodamide product will be ______.
Concept: undefined >> undefined
Hydrogen has three isotopes: protium (1H), deuterium (2H or D) and tritium (3H or T). They have nearly the same chemical properties but different physical properties. They differ in ______.
Concept: undefined >> undefined
Which one of the following statements for D.I. Mendeleev is incorrect?
Concept: undefined >> undefined
Given below are two statements, one is labelled as Assertion (A) and the other is labelled as Reason (R).
Assertion (A) : Lithium salts are hydrated.
Reason (R) : Lithium has higher polarising power than other alkali metal group members.
In the light of the above statements, choose the most appropriate answer from the options given below :
Concept: undefined >> undefined
The correct order of conductivity of ions in water is ______.
Concept: undefined >> undefined
KO2 (potassium super oxide) is used in oxygen cylinders in space and submarines because it ______.
Concept: undefined >> undefined
The acid that is believed to be mainly responsible for the damage of Taj Mahal is ______.
Concept: undefined >> undefined
Given below are two statements:
Statement I: Phenols are weekly acidic.
Statement II: Therefore they are freely soluble in NaOH solutions and are weaker acids than alcohols and water.
Choose the most appropriate options:
Concept: undefined >> undefined
(A) \[\ce{HOCl + H2O2 -> H3O^+ + Cl^- + O2}\]
(B) \[\ce{I2 + H2O2 + 2OH^- -> 2I^- + 2H2O + O2}\]
Choose the correct option.
Concept: undefined >> undefined
Given below are two statements:
Statement I: The E° value for \[\ce{\frac{Ce^{4+}}{Ce^{3+}}}\] is +1.74 V.
Statement II: Ce is more stable in Ce4+ state than Ce3+ state.
In the light of the above statements, choose the correct answer from the options given below:
Concept: undefined >> undefined
\[\ce{2MnO^-4 + {b}C2O^{2-}4 + {c}H^+ -> {x}Mn^{2+} + {y}CO2 + {z}H2O}\]
If the above equation is balanced with integer coefficients, the value of c is ______. (Round off to the Nearest Integer).
Concept: undefined >> undefined
Which one of the following when dissolved in water gives coloured solution in nitrogen atmosphere?
Concept: undefined >> undefined
The correct options for the products A and B of the following reactions are:
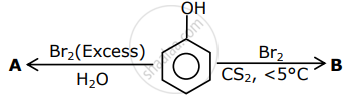
Concept: undefined >> undefined
The ratio of number of water molecules in Mohr's salt and potash alum is ______ × 10-1.
Concept: undefined >> undefined
