Advertisements
Advertisements
Match the substances in Column I with those in Column II.
| Column 1 | Column II | ||
| (a) | Tartaric acid | (i) | soap |
| (b) | Calcium hydroxide | (ii) | curd |
| (c) | Formic acid | (iii) | unripe mangoes |
| (d) | Sodium hydroxide | (iv) | ant’s sting |
| (e) | Lactic acid | (v) | lime water |
Concept: undefined >> undefined
Lemon juice and vinegar taste ______ because they contain ______.
Concept: undefined >> undefined
Advertisements
When an acidic solution is mixed with a basic solution, they ______ each other forming ______ and water.
Concept: undefined >> undefined
Fill in the cross word given in figure with the help of the clues provided.
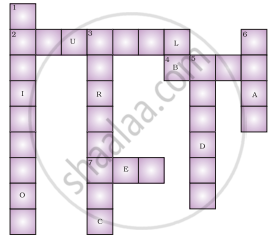
Across
(2) The solution which does not change the colour of either red or blue litmus.
(4) Phenolphthalein gives pink colour in this type of solution.
(7) Colour of blue litmus in lemon juice.
Down
(1) It is used to test whether a substance is acidic or basic.
(3) It is a natural indicator and gives pink colour in basic solution.
(5) Nature of ant’s sting.
(6) It is responsible for the increase in temperature during a neutralisation reaction.
Concept: undefined >> undefined
A farmer was unhappy because of his low crop yield. He discussed the problem with an agricultural scientist and realised that the soil of his field was either too acidic or too basic. What remedy would you suggest the farmer to neutralise the soil?
Concept: undefined >> undefined
You are provided with four test tubes containing sugar solution, baking soda solution, tamarind solution, salt solution. Write down an activity to find the nature (acidic/basic/neutral) of each solution.
Concept: undefined >> undefined
Paheli observed that most of the fish in the pond of her village were gradually dying. She also observed that the waste of a factory in their village is flowing into the pond which probably caused the fish to die.
- Explain why the fish were dying.
- If the factory waste is acidic in nature, how can it be neutralised?
Concept: undefined >> undefined
Which of the following is a physical change?
Concept: undefined >> undefined
Paheli’s mother made a concentrated sugar syrup by dissolving sugar in hot water. On cooling, crystals of sugar got separated. This indicates a –
Concept: undefined >> undefined
Two drops of dilute sulphuric acid were added to 1 g of copper sulphate powder and then small amount of hot water was added to dissolve it (step I). On cooling, beautiful blue coloured crystals got separated (step II). Step I and Step II are:
Concept: undefined >> undefined
State whether the following statement is true or false:
Anaerobic bacteria digest animal waste and produce biogas.
Concept: undefined >> undefined
State whether the following statement is true or false:
Stretching of rubber bands is not a physical change.
Concept: undefined >> undefined
What kind of change is shown by tearing of paper?
Concept: undefined >> undefined
Match the items of Column I with the items of Column II.
| Column I | Column II | ||
| (a) | Large crystals | (i) | Turns lime water into milky |
| (b) | Depositing a layer of zinc on iron | (ii) | Physical change |
| (c) | Souring of milk | (iii) | Rust |
| (d) | Carbon dioxide | (iv) | Sugar candy (Mishri) |
| (e) | Iron oxide | (v) | Chemical change |
| (f) | Dissolving common salt in water | (vi) | Galvanisation |
Concept: undefined >> undefined
Fill in the blanks in the following statements using the words given in the box.
| rusted, colourful, substance, chemical, physical, reversible, iron oxide, object |
- Making sugar solution is a ______ change.
- A physical change is generally ______.
- Grinding of wheat grain changes its size. It is a ______ change.
- Iron benches kept in lawns and gardens get ______. It is a ______ change because a new ______ is formed.
Concept: undefined >> undefined
A physical change is generally____________.
Concept: undefined >> undefined
Grinding of wheat grain changes its size. It is a ____________ change.
Concept: undefined >> undefined
Give two examples for the following case:
Physical changes which are reversible
Concept: undefined >> undefined
Give two examples for the following case:
Physical changes which are not reversible
Concept: undefined >> undefined
If you leave a piece of iron in the open for a few days, it acquires a film of brownish substance, called rust.
Do you think rust is different from iron?
Concept: undefined >> undefined
