Advertisements
Advertisements
Write the expression for the de Broglie wavelength associated with a charged particle of charge q and mass m, when it is accelerated through a potential V.
Concept: undefined >> undefined
State de Broglie hypothesis.
Concept: undefined >> undefined
Advertisements
Why we do not see the wave properties of a baseball?
Concept: undefined >> undefined
A proton and an electron have the same kinetic energy. Which one has a greater de Broglie wavelength? Justify.
Concept: undefined >> undefined
Write the relationship of de Broglie wavelength λ associated with a particle of mass m in terms of its kinetic energy K.
Concept: undefined >> undefined
An electron and an alpha particle have the same kinetic energy. How are the de Broglie wavelengths associated with them related?
Concept: undefined >> undefined
What is Bremsstrahlung?
Concept: undefined >> undefined
Explain why photoelectric effect cannot be explained on the basis of wave nature of light.
Concept: undefined >> undefined
Derive an expression for de Broglie wavelength of electrons.
Concept: undefined >> undefined
A beam of light consisting of red, green and blue is incident on a right-angled prism as shown in figure. The refractive index of the material of the prism for the above red, green and blue colours are 1.39, 1.44 and 1.47 respectively. What are the colours suffer total internal reflection?
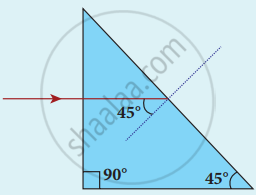
Concept: undefined >> undefined
Briefly explain the principle and working of electron microscope.
Concept: undefined >> undefined
Describe briefly Davisson – Germer experiment which demonstrated the wave nature of electrons.
Concept: undefined >> undefined
How do we obtain characteristic x-ray spectra?
Concept: undefined >> undefined
What should be the velocity of the electron so that its momentum equals that of 4000 Å wavelength photon.
Concept: undefined >> undefined
Calculate the de Broglie wavelength of a proton whose kinetic energy is equal to 81.9 × 10–15 J.
(Given: mass of proton is 1836 times that of electron).
Concept: undefined >> undefined
A deuteron and an alpha particle are accelerated with the same potential. Which one of the two has
- greater value of de Broglie wavelength associated with it and
- less kinetic energy?
Explain.
Concept: undefined >> undefined
An electron is accelerated through a potential difference of 81 V. What is the de Broglie wavelength associated with it? To which part of the electromagnetic spectrum does this wavelength correspond?
Concept: undefined >> undefined
The ratio between the de Broglie wavelength associated with proton accelerated through a potential of 512 V and that of alpha particle accelerated through a potential of X volts is found to be one. Find the value of X.
Concept: undefined >> undefined
The barrier potential of a silicon diode is approximately, ____________.
Concept: undefined >> undefined
In an unbiased p-n junction, the majority of charge carriers (that is, holes) in the p-region diffuse into the n-region because of
Concept: undefined >> undefined
