Advertisements
Advertisements
Which one of the following substituents at para-position is most effective in stabilizing the phenoxide 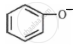 ion?
ion?
Concept: undefined >> undefined
Which one of the following reactions will not result in the formation of a carbon-carbon bond?
Concept: undefined >> undefined
Advertisements
Which of the following compounds will undergo self-aldol condensation in the presence of cold dilute alkali?
Concept: undefined >> undefined
The amine, which reacts with p-toluenesulphonyl chloride to give a clear solution, which on acidification gives insoluble compound is ______.
Concept: undefined >> undefined
Which of the following compound will give positive iodoform test?
Concept: undefined >> undefined
A substance C4H10O yields on oxidation a compound, C4H8O which gives an oxime and a positive iodoform test. The original substance on treatment with a conc. H2SO4 gives C4H8. The structure of the compound is ______.
Concept: undefined >> undefined
The amino acid, which is basic in nature is ______.
Concept: undefined >> undefined
Which among the following enzymes hydrolysis starch?
Concept: undefined >> undefined

In the above reaction, product (B) is:
Concept: undefined >> undefined
Calculate ΔU if 2 kJ heat is released and 10 kJ of work is done on the system.
Concept: undefined >> undefined
Which of the following reaction does not involve either oxidation or reduction?
Concept: undefined >> undefined
Which symbol replaces the unit of atomic mass, amu?
Concept: undefined >> undefined
Which of the following is most reactive towards the addition reaction of hydrogen cyanide to form the corresponding cyanohydrin?
Concept: undefined >> undefined
What is the number of different products formed when mixture of ethanal and propanal reacts with aq. NaOH after warming?
Concept: undefined >> undefined
Which of the following compounds does not undergo a haloform reaction?
Concept: undefined >> undefined
Calculate the work done during the combustion of 0.138 kg of ethanol, C2H5OH(l) at 300 K.
Given: R = 8.314 Jk−1 mol−1, molar mass of ethanol = 46 g mol−1.
Concept: undefined >> undefined
Tertiary structure of protein is stabilised by ______.
Concept: undefined >> undefined
Which of the following is not correctly matched?
Concept: undefined >> undefined
0.05 F electricity is passed through CuSO4 solution. Calculate the mass of Cu produced at cathode? (molar mass of Cu = 63.5 g mol-1)
Concept: undefined >> undefined
Identify the reaction in which carbonyl group of aldehydes and ketones is reduced to methylene group on treatment with zinc-amalgam and cone. HCI.
Concept: undefined >> undefined
