Advertisements
Advertisements
Which of the following is a hydrogen bonded molecular solid?
Concept: undefined >> undefined
Calcite and ____________ are two forms of calcium carbonate.
Concept: undefined >> undefined
Advertisements
If \[\ce{C(s) + O2(g) -> CO2(g), \Delta H = - \text{x}}\]
\[\ce{CO(g) + \frac{1}{2} O2(g) -> CO2(g), \Delta H = - \text{y}}\],
Calculate Δf H for CO(g) formation.
Concept: undefined >> undefined
Which among the following compounds is obtained when ethane nitrile is acid hydrolysed?
Concept: undefined >> undefined
For the reaction \[\ce{A + 2B -> C}\], the instantaneous rate of reaction at a given instant can be represented by ____________.
Concept: undefined >> undefined
Sea water is a homogeneous mixture of ____________ type of solution.
Concept: undefined >> undefined
Which of the following molecules have London forces as predominant intermolecular force of attraction?
Concept: undefined >> undefined
The following structure can be represented as:
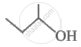
Concept: undefined >> undefined
In which of the following, the central atom does NOT have lone pair(s) of electrons?
Concept: undefined >> undefined
The general electronic configuration of group 17 elements is ____________.
Concept: undefined >> undefined
[EDTA]4− and oxalate ions are examples of ____________ ligands respectively.
Concept: undefined >> undefined
The compound incapable of hydrogen bonding with water is ______.
Concept: undefined >> undefined
In wedge formula, the solid wedge is used to indicate ____________.
Concept: undefined >> undefined
Which of the following is CORRECT about the two allotropic forms of polonium?
Concept: undefined >> undefined
In ____________, hydrogen bonding is stronger than corresponding alcohols.
Concept: undefined >> undefined
The conversion of 2-methylpropan-1-ol to 2-methylpropan-2-ol is ______.
Concept: undefined >> undefined
In a reaction \[\ce{2A + P -> A2P}\], the reactant 'A' disappears at ____________.
Concept: undefined >> undefined
What is the IUPAC name of α-methyl butyraldehyde?
Concept: undefined >> undefined
The condensed electronic configuration [Ne] 3s2 3p5 denotes the element ____________.
Concept: undefined >> undefined
The rate of disappearance of SO2 in the reaction \[\ce{2SO2 + O2 -> 2SO3}\] is 1.28 × 10−3 mol dm−3 s−1. Then the rate of formation of SO3 is ____________ mol dm−3 s−1.
Concept: undefined >> undefined
