Advertisements
Advertisements
If liquids A and B form an ideal solution, the ______.
Concept: undefined >> undefined
For a reaction, given below is the graph of ln k vs `1/"T"`. The activation energy for the reaction is equal to ______ cal mol-1. (Nearest integer) (Given : R = 2 cal K-1 mol-1)

Concept: undefined >> undefined
Advertisements
Which one of the following statements is not true about enzymes?
Concept: undefined >> undefined
Which of the following chemical reactions represents Hall- Heroult Process?
Concept: undefined >> undefined
The purest form of commercial iron is ______.
Concept: undefined >> undefined
Which of the following is the most stable carbocation?
Concept: undefined >> undefined
Among the following statements on the nitration of aromatic compounds, the false statement is:
Concept: undefined >> undefined
The energy of an electron in the first Bohr orbit of the H-atom is −13.6 eV. The energy value of an electron in the excited state of Li2+ is ______.
Concept: undefined >> undefined
Which one of the following has the largest ionic radius?
Concept: undefined >> undefined
Low oxidation state of metals in their complexes are common when ligands ______.
Concept: undefined >> undefined
Which will have the highest enol content?
Concept: undefined >> undefined
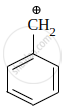 |
\[\ce{CH2 = \overset{⊕}{C}H}\] | \[\ce{CH3 - \overset{⊕}{C}H2}\] | \[\ce{HC ≡ \overset{⊕}{C}}\] |
| A | B | C | D |
The correct order of stability of given carbocation is:
Concept: undefined >> undefined
A solution of (–) – 1 – chloro–1–phenylethane in toluene racemises slowly in the presence of a small amount of SbCl5, due to the formation of ______.
Concept: undefined >> undefined
The increasing order of stability of the following free radicals is ______.
Concept: undefined >> undefined
In a hydrogen-oxygen fuel cell, combustion of hydrogen occurs to ______.
Concept: undefined >> undefined
In general, the property (magnitudes only) that shows an opposite trend in comparison to other properties across a period is ______.
Concept: undefined >> undefined
Compound A is converted to B on reaction with CHCl3 and KOH. The compound B is toxic and can be decomposed by C. A, B and C respectively are ______.
Concept: undefined >> undefined
Consider the elements Mg, Al, S, P and Si, the correct increasing order of their first ionization enthalpy is ______.
Concept: undefined >> undefined
The correct order of intensity of colors of the compounds is ______.
Concept: undefined >> undefined
An inorganic compound 'X' on treatment with concentrated H2SO4 produces brown fumes and gives a dark brown ring with FeSO4 in presence of concentrated H2SO4. Also, compound 'X' gives precipitate 'Y', when its solution in dilute HCl is treated with H2S gas. The precipitate 'Y' on treatment with concentrated HNO3 followed by an excess of NH4OH further gives a deep blue coloured solution, compound 'X' is:
Concept: undefined >> undefined
