Advertisements
Advertisements
An exothermic reaction X → Y has an activation energy 30 kJ mol-1. If energy change ΔE during the reaction is - 20 kJ, then the activation energy for the reverse reaction in kJ is ______.
Concept: undefined >> undefined
A schematic plot of ln Keq versus inverse of temperature for a reaction is shown below

The reaction must be:
Concept: undefined >> undefined
Advertisements
Assertion: For the extraction of iron, haematite ore is used.
Reason: Haematite is a carbonate ore of iron.
Concept: undefined >> undefined
Which of the following is incorrect?
Concept: undefined >> undefined
\[\ce{Au + CN^- + H2O + O2 -> [Au(CN)2]^- + OH^-}\]
The number of CN− ions involved in the balanced equation is ______.
Concept: undefined >> undefined
Heat treatment of muscular pain involves radiation of a wavelength of about 900 nm. Which spectral line of H-atom is suitable for this purpose?
[RH = 1 × 105 cm−1, h = 6.6 × 10−34 Js, c = 3 × 108 ms−1]
Concept: undefined >> undefined
Which one of the following chemicals have not been used for the dry cleaning of clothes?
Concept: undefined >> undefined
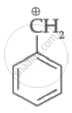
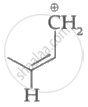
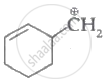
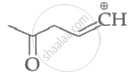
Which of the following carbocation's is most stable?
Concept: undefined >> undefined
 |
 |
 |
 |
| (I) | (II) | (III) | (IV) |
Among the given species the resonance stabilised carbocations are:
Concept: undefined >> undefined
Al2O3 was leached with alkali to get X. The solution of X on passing of gas Y forms Z. X, Y and Z respectively are:
Concept: undefined >> undefined
Match List-I with List-II:
| List-I | List-II |
| Industrial process | Application |
| (a) Haber's process | (i) HNO3 synthesis |
| (b) Ostwald's process | (ii) Aluminium extraction |
| (c) Contact process | (iii) NH3 synthesis |
| (d) Hall-Heroult process | (iv) H2SO4 synthesis |
Choose the correct answer from the options given below:
Concept: undefined >> undefined
Given below are two statements:
Statements I: According to Bohr's model of an atom, qualitatively the magnitude of velocity of electron increases with decrease in positive charges on the nucleus as there is no strong hold on the electron by the nucleus.
Statement II: According to Bohr's model of an atom, qualitatively the magnitude of velocity of electron increase with a decrease in principal quantum number.
In light of the above statements, choose the most appropriate answer from the options given below:
Concept: undefined >> undefined
While titration dilute HCl solution with aqueous NaOH, which of the following will not be required?
Concept: undefined >> undefined
The size of iso-electronic species Cl-, Ar and Ca2+ is affected by ______.
Concept: undefined >> undefined
The wavelength in Å of the photon that is emitted when an electron in Bohr orbit with n = 2 returns to orbit with n = 1 in H atom is ______ Å. The ionisation potential of the ground state of the H-atom is 2.17 × 10−11 erg.
Concept: undefined >> undefined
Benzoic acid gives benzene on being heated with X and phenol gives benzene on being heated with Y. Therefore X and Y are respectively:
Concept: undefined >> undefined
The number of times larger the spacing between the energy levels with n = 3 and n = 8 spacing between the energy level with n = 8 and n = 9 for the hydrogen atom is ______.
Concept: undefined >> undefined
The first ionization energy of H is 21.79 × 10-19 J. The second ionization energy of He atom is ______ × 10-19J.
Concept: undefined >> undefined
The line at 434 nm in the Balmer series of the hydrogen spectrum corresponds to a transition of an electron from the nth to second Bohr orbit. The value of n is ______.
Concept: undefined >> undefined
