Advertisements
Advertisements
In the given reactions sequence, the major product 'C' is:
\[\ce{C8H10 ->[HNO3][H2SO4] A ->[Br2][\Delta] B ->[alcoholic][KOH] C}\]
Concept: undefined >> undefined
The decreasing order of reactivity of the following organic molecules towards AgNO3 solution is ______.
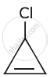

- \[\begin{array}{cc}\ce{CH3CHCH3}\\
|\phantom{..}\\\ce{Cl}\phantom{.}\end{array}\] - \[\begin{array}{cc}\ce{CH3CHCH2NO2}\\
|\phantom{......}\\\ce{Cl}\phantom{.....}\end{array}\]
Concept: undefined >> undefined
Advertisements
The number of stereoisomers are possible for a compound of the molecular formula \[\ce{CH3 - CH = CH - CH(OH) - Me}\] is ______.
Concept: undefined >> undefined
Arrange the following in increasing order of reactivity towards nitration
- p-xylene
- bromobenzene
- mesitylene
- nitrobenzene
- benzene
Choose the correct answer from the options given below:
Concept: undefined >> undefined
Number of amphoteric compound among the following.
Concept: undefined >> undefined
- Phenyl methenamine
- N, N - Dimethylaniline
- N - Methyl aniline
- Benzenamine
Choose the correct order of the basic nature of the above amines.
Concept: undefined >> undefined
The mechanism of action of "Terfenadine" (Seldane) is ______.
Concept: undefined >> undefined
The amine 'A' when treated with nitrous acid gives yellow oily substance. The amine A is ______.
Concept: undefined >> undefined
Which of the following statement(s) is/are incorrect in case of Hofmann bromamide degradation?
Concept: undefined >> undefined
The polymer, which can be stretched and retains its original status on releasing the force is ______.
Concept: undefined >> undefined
An acidified manganate solution undergoes disproportionation reaction. The spin-only magnetic moment value of the product having manganese in a higher oxidation state is ______ B.M. (Nearest integer)
Concept: undefined >> undefined
Which of the following are isostructural pairs?
(A) \[\ce{SO^{2-}4}\] and \[\ce{CrO^{2-}4}\]
(B) SiCl4 and TiCl4
(C) NH3 and \[\ce{NO^-3}\]
(D) BCl3 and BrCl3
Concept: undefined >> undefined
The reaction of sulphur in alkaline medium is given below:
\[\ce{S8(s) + {a} OH^-(aq) -> {b} S^{2-}(aq) + {c} S2O^{2-}3(aq) + {d} H2O(l)}\]
The values of 'a' are ______. (Integer answer)
Concept: undefined >> undefined
The species given below that does NOT show a disproportionation reaction is ______.
Concept: undefined >> undefined
To synthesise 1.0 mole of 2-methylpropan-2-ol from Ethylethanoate ______ equivalents of CH3MgBr reagent will be required. (Integer value)
Concept: undefined >> undefined


The products "A" and "B" formed in above reactions are:
Concept: undefined >> undefined
Consider the following reactions:
(i) \[\ce{Glucose + R-OH ->[Conc. HNO3] [A] ->[X eq of][(CH3CO)2O] Acetyl derivative}\]
(ii) \[\ce{Glucose ->[Ni/H2] [A] ->[Y eq of][(CH3CO)2O] Acetyl derivative}\]
(iii) \[\ce{Glucose ->[Z eq of][(CH3CO)2O] Acetyl derivative}\]
'X, 'Y' and 'Z' in these reactions are respectively:
Concept: undefined >> undefined
An aldehyde isomeric with allyl alcohol gives phenyl hydrazone. Pick out a ketone that too gives a phenyl hydrazone containing the same percentage of nitrogen.
Concept: undefined >> undefined
\[\ce{C3H8O ->[{[O]}][K2Cr2O7/H2SO4] C3H6O ->[I2 + NaOH(aq.)] CHI3}\]
In this reaction the first compound is:
Concept: undefined >> undefined
\[\ce{H2O2 -> H2O + O2}\]
This represents ______.
Concept: undefined >> undefined
