Advertisements
Advertisements
Write the main product in the following reaction:
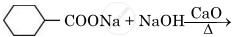
Concept: undefined >> undefined
DNA fingerprinting is used to determine paternity of an individual. Which property of DNA helps in the procedure?
Concept: undefined >> undefined
Advertisements
Account for the following:
Fluorine forms only one oxoacid HOF.
Concept: undefined >> undefined
Draw the structures of the following:
(1) HClO4
(2) H3PO3
Concept: undefined >> undefined
When RNA is hydrolysed, there is no relationship among the quantities of different bases obtained. What does this fact suggest about the structure of RNA?
Concept: undefined >> undefined
Draw the structures of the HClO3.
Concept: undefined >> undefined
Account for the following :
HClO4 is a stronger acid than HClO.
Concept: undefined >> undefined
The increasing order of reducing power of the halogen acids is:
Concept: undefined >> undefined
Which of the following statements is true for protein synthesis?
Concept: undefined >> undefined
Which one of the following order is correct for the bond dissociation enthalpy of halogen molecule?
Concept: undefined >> undefined
Assertion: HI cannot be prepared by the reaction of KI with concentrated H2SO4.
Reason: HI has the lowest H-X bond strength among halogen acids.
Concept: undefined >> undefined
A black compound of manganese reacts with a halogen acid to give greenish-yellow gas. When excess of this gas reacts with \[\ce{NH3}\] an unstable trihalide is formed. In this process the oxidation state of nitrogen changes from ______.
Concept: undefined >> undefined
Explain why the stability of oxoacids of chlorine increases in the order given below:
\[\ce{HClO < HClO2 < HClO3 < HClO4}\]
Concept: undefined >> undefined
Assertion: \[\ce{HI}\] cannot be prepared by the reaction of KI with concentrated \[\ce{H2SO4}\]
Reason: \[\ce{HI}\] has lowest \[\ce{H - X}\] bond strength among halogen acids.
Concept: undefined >> undefined
Draw the structures of the following molecules: BrF3
Concept: undefined >> undefined
ICl is more reactive than I2.
Concept: undefined >> undefined
Account for the following: Interhalogens are more reactive than pure halogens.
Concept: undefined >> undefined
Draw the structure of BrF5
Concept: undefined >> undefined
Draw the structures of the following : ClF3
Concept: undefined >> undefined
Why is ICl more reactive than I2?
Concept: undefined >> undefined
