Advertisements
Advertisements
An organic molecule necessarily shows optical activity if it ____________.
Concept: undefined >> undefined
Which of the following is a chiral compound?
Concept: undefined >> undefined
Advertisements
Among the following, the dissociation constant is highest for:
Concept: undefined >> undefined
Which of the following is a disaccharide?
Concept: undefined >> undefined
The sugar that is characteristic of milk is ____________.
Concept: undefined >> undefined
Which one is a disaccharide?
Concept: undefined >> undefined
The commonest disaccharide has the molecular formula:
Concept: undefined >> undefined
Maltose and glucose are ____________.
Concept: undefined >> undefined
Sucrose on hydrolysis gives:
Concept: undefined >> undefined
The boiling point of an azeotropic mixture of water and ethanol is less than that of water and ethanol. The mixture shows ____________.
Concept: undefined >> undefined
SN1 reaction of alkyl halides lead to ___________.
Concept: undefined >> undefined
Identify the end product (C) in the following sequence:
\[\ce{C2H5OH ->[SOCl2][Pyridine] A ->[KCN {(alc.)}] B ->[2H2O/H^+] C}\]
Concept: undefined >> undefined
Identify X and Y in the following sequence:
\[\ce{C2H5Br ->[X] Product ->[Y] C3H7NH2}\]
Concept: undefined >> undefined
Which of the following has not a glucosidic linkage?
Concept: undefined >> undefined
The coordination number of Y will be in the XY types of crystal:
Concept: undefined >> undefined
Considering the formation, breaking and strength of hydrogen bond, predict which of the following mixtures will show a positive deviation from Raoult’s law?
Concept: undefined >> undefined
Which of the following undergoes nucleophilic substitution exclusively by SN1 mechanism?
Concept: undefined >> undefined
The increasing order of nucleophilicity would be:
Concept: undefined >> undefined
The correct order of increasing the reactivity of C–X bond towards nucleophile in following compounds.

(I)

(II)
(CH3)3CCl
(III)
(CH3)2CHCl
(IV)
Concept: undefined >> undefined
Which of the following compound will undergo racemisation when reacts with aq. KOH?
(i)
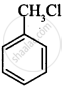
(ii)
CH3CH2CH2Cl
(iii)
\[\begin{array}{cc}
\ce{CH3}\phantom{..}\\
|\phantom{....}\\
\ce{CH3-CH-CH2Cl}
\end{array}\]
(iv)
\[\begin{array}{cc}
\phantom{..}\ce{H}\\
\phantom{..}|\\
\ce{CH3-C-Cl}\\
\phantom{..}|\\
\phantom{.....}\ce{C2H5}
\end{array}\]
Concept: undefined >> undefined
