Advertisements
Advertisements
Explain why the following system is not aromatic?

Concept: undefined >> undefined
Explain why the following system is not aromatic?

Concept: undefined >> undefined
Advertisements
The ionization constant of propanoic acid is 1.32 × 10–5. Calculate the degree of ionization of the acid in its 0.05M solution and also its pH. What will be its degree of ionization if the solution is 0.01M in HCl also?
Concept: undefined >> undefined
One mole of any substance contains 6.022 × 1023 atoms/molecules. Number of molecules of \[\ce{H2SO4}\] present in 100 mL of 0.02 M \[\ce{H2SO4}\] solution is ______.
Concept: undefined >> undefined
Which of the following statements about a compound is incorrect?
Concept: undefined >> undefined
16 g of oxygen has same number of molecules as in:
(i) \[\ce{16 g of CO}\]
(ii) \[\ce{28 g of N2}\]
(iii) \[\ce{14 g of N2}\]
(iv) \[\ce{1.0 g of H2}\]
Concept: undefined >> undefined
The entropy change can be calculated by using the expression ∆S = `q_(rev)/T`. When water freezes in a glass beaker, choose the correct statement amongst the following :
Concept: undefined >> undefined
The spontaneity means, having the potential to proceed without the assistance of external agency. The processes which occur spontaneously are:
(i) Flow of heat from colder to warmer body.
(ii) Gas in a container contracting into one corner.
(iii) Gas expanding to fill the available volume.
(iv) Burning carbon in oxygen to give carbon dioxide.
Concept: undefined >> undefined
Enthalpy diagram for a particular reaction is given in figure. Is it possible to decide spontaneity of a reaction from given diagram. Explain.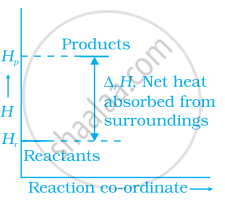
Concept: undefined >> undefined
Match the following processes with entropy change:
| Reaction | Entropy change |
| (i) A liquid vapourises | (a) ∆S = 0 |
| (ii) Reaction is non-spontaneous at all temperatures and ∆H is positive |
(b) ∆S = positive |
| (iii) Reversible expansion of an ideal gas | (c) ∆S = negative |
Concept: undefined >> undefined
Assertion (A): A liquid crystallises into a solid and is accompanied by decrease in entropy.
Reason (R): In crystals, molecules organise in an ordered manner.
Concept: undefined >> undefined
Four structures are given in options (i) to (iv). Examine them and select the aromatic structures.
| (i) |  |
| (ii) |  |
| (iii) |  |
| (iv) |  |
Concept: undefined >> undefined
Write the electronic configurations of the given ions H–.
Concept: undefined >> undefined
Write the electronic configurations of the given ions Na+.
Concept: undefined >> undefined
Write the electronic configurations of the given ions O2–.
Concept: undefined >> undefined
Write the electronic configurations of the given ions F–.
Concept: undefined >> undefined
What is the lowest value of n that allows g orbitals to exist?
Concept: undefined >> undefined
An Atom of an Element Contains 29 Electrons and 35 Neutrons. Deduce
(i) the Number of Protons and
(ii) the Electronic Configuration of the Element.
Concept: undefined >> undefined
Give the number of electrons in the species `"H"_2^+`, H2 and `"O"_2^+`
Concept: undefined >> undefined
A certain particle carries 2.5 × 10-16 C of static electric charge. Calculate the number of electrons present in it.
Concept: undefined >> undefined
