Advertisements
Advertisements
Isotherms of carbon dioxide gas are shown in figure. Mark a path for changing gas into liquid such that only one phase (i.e., either a gas or a liquid) exists at any time during the change. Explain how the temperature, volume and pressure should be changed to carry out the change.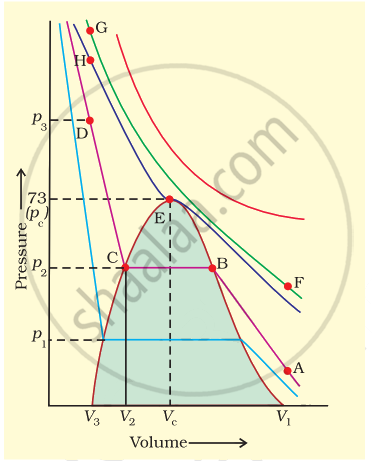
Concept: undefined >> undefined
Enthalpy is an extensive property. In general, if enthalpy of an overall reaction A→B along one route is ∆rH and ∆rH1, ∆rH2, ∆rH3 ..... represent enthalpies of intermediate reactions leading to product B. What will be the relation between ∆rH for overall reaction and ∆rH1, ∆rH2 ..... etc. for intermediate reactions.
Concept: undefined >> undefined
Advertisements
The difference between CP and CV can be derived using the empirical relation H = U + pV. Calculate the difference between CP and CV for 10 moles of an ideal gas.
Concept: undefined >> undefined
The enthalpy of vapourisation of \[\ce{CCl4}\] is 30.5 kJ mol–1. Calculate the heat required for the vapourisation of 284 g of \[\ce{CCl4}\] at constant pressure. (Molar mass of \[\ce{CCl4}\] = 154 g mol–1).
Concept: undefined >> undefined
Draw the gas phase and solid phase structure of \[\ce{H2O2}\].
Concept: undefined >> undefined
Write the Lewis structure of hydrogen peroxide.
Concept: undefined >> undefined
How will you concentrate \[\ce{H2O2}\]? Show differences between structures of \[\ce{H2O2}\] and \[\ce{H2O}\] by drawing their spatial structures. Also mention three important uses of \[\ce{H2O2}\].
Concept: undefined >> undefined
A colourless liquid ‘A’ contains H and O elements only. It decomposes slowly on exposure to light. It is stabilised by mixing urea to store in the presence of light. Suggest possible structure of A.
Concept: undefined >> undefined
An alkane C8H18 is obtained as the only product on subjecting a primary alkyl halide to Wurtz reaction. On monobromination this alkane yields a single isomer of a tertiary bromide. Write the structure of alkane and the tertiary bromide.
Concept: undefined >> undefined
Use Bohr's postulate to prove that the radius of nth orbit in a hydrogen atom is proportional to n2.
Concept: undefined >> undefined
The increasing order of reactivity among group 1 elements is Li < Na < K < Rb <Cs whereas that among group 17 elements is F > CI > Br > I. Explain.
Concept: undefined >> undefined
The first (Δ1H1) and the second (Δ1H2) ionization enthalpies (in kJ mol-1) and the (ΔegH) electron gain enthalpy (in kJ mol-1) of a few elements are given below:
| Elements | Δ1H1 | Δ1H2 | ΔegH |
| I | 520 | 7300 | -60 |
| II | 419 | 3051 | -48 |
| III | 1681 | 3374 | -328 |
| IV | 1008 | 1846 | -295 |
| V | 2372 | 5251 | +48 |
| VI | 738 | 1451 | -40 |
Which of the above elements is likely to be the least reactive element.
Concept: undefined >> undefined
The first (Δ1H1) and the second (Δ1H2) ionization enthalpies (in kJ mol-1) and the (ΔegH) electron gain enthalpy (in kJ mol-1) of a few elements are given below:
| Elements | Δ1H1 | Δ1H2 | ΔegH |
| I | 520 | 7300 | -60 |
| II | 419 | 3051 | -48 |
| III | 1681 | 3374 | -328 |
| IV | 1008 | 1846 | -295 |
| V | 2372 | 5251 | +48 |
| VI | 738 | 1451 | -40 |
Which of the above elements is likely to be the most reactive metal.
Concept: undefined >> undefined
The first (Δ1H1) and the second (Δ1H2) ionization enthalpies (in kJ mol-1) and the (ΔegH) electron gain enthalpy (in kJ mol-1) of a few elements are given below:
| Elements | Δ1H1 | Δ1H2 | ΔegH |
| I | 520 | 7300 | -60 |
| II | 419 | 3051 | -48 |
| III | 1681 | 3374 | -328 |
| IV | 1008 | 1846 | -295 |
| V | 2372 | 5251 | +48 |
| VI | 738 | 1451 | -40 |
Which of the above elements is likely to be the most reactive non-metal.
Concept: undefined >> undefined
The first (Δ1H1) and the second (Δ1H2) ionization enthalpies (in kJ mol-1) and the (ΔegH) electron gain enthalpy (in kJ mol-1) of a few elements are given below:
| Elements | Δ1H1 | Δ1H2 | ΔegH |
| I | 520 | 7300 | -60 |
| II | 419 | 3051 | -48 |
| III | 1681 | 3374 | -328 |
| IV | 1008 | 1846 | -295 |
| V | 2372 | 5251 | +48 |
| VI | 738 | 1451 | -40 |
Which of the above elements is likely to be the least reactive non-metal.
Concept: undefined >> undefined
The first (Δ1H1) and the second (Δ1H2) ionization enthalpies (in kJ mol-1) and the (ΔegH) electron gain enthalpy (in kJ mol-1) of a few elements are given below:
| Elements | Δ1H1 | Δ1H2 | ΔegH |
| I | 520 | 7300 | -60 |
| II | 419 | 3051 | -48 |
| III | 1681 | 3374 | -328 |
| IV | 1008 | 1846 | -295 |
| V | 2372 | 5251 | +48 |
| VI | 738 | 1451 | -40 |
Which of the above elements is likely to be the metal which can form a stable binary halide of the formula MX2, (X=halogen).
Concept: undefined >> undefined
The first (Δ1H1) and the second (Δ1H2) ionization enthalpies (in kJ mol-1) and the (ΔegH) electron gain enthalpy (in kJ mol-1) of a few elements are given below:
| Elements | Δ1H1 | Δ1H2 | ΔegH |
| I | 520 | 7300 | -60 |
| II | 419 | 3051 | -48 |
| III | 1681 | 3374 | -328 |
| IV | 1008 | 1846 | -295 |
| V | 2372 | 5251 | +48 |
| VI | 738 | 1451 | -40 |
Which of the above elements is likely to bethe metal which can form a predominantly stable covalent halide of the formula MX (X = halogen)?
Concept: undefined >> undefined
Considering the elements F, Cl, O and N, the correct order of their chemical reactivity in terms of oxidizing property is ______.
Concept: undefined >> undefined
Calculate the wavelength of an electron moving with a velocity of 2.05 × 107 ms-1.
Concept: undefined >> undefined
The mass of an electron is 9.1 × 10–31 kg. If its K.E. is 3.0 × 10–25 J, calculate its wavelength.
Concept: undefined >> undefined
